Frederick Reif
(1927-2019)
Frederick "Fred" Reif earned his Ph.D in physics from Harvard University in 1953, with Ed Purcell as his thesis adviser. He went to work with Enrico Fermi at the University of Chicago. Seven years later, he went to the University of California, Berkeley, where he taught for twenty-nine years. At Berkeley he authored a classic text in statistical physics and thermodynamics,
Statistical and Thermal Physics, published in 1965.
At that time, the National Science Foundation supported the development of more modern textbook series in the physical sciences aimed at high schools students. At MIT, Jerrold Zacharias led the
Physical Science Study Committee. At Harvard, Gerald Holton and Fletcher Watson led
Project Physics. At Berkeley, Reif rewrote his textbook, titled simply
Statistical Physics, as volume 5 of the
Berkeley Physics Course.
This book incorporated the first sophisticated computer calculations of the motions of gas molecules. As such, it was a landmark in the field of computational methods of solving problems in physics as opposed to solving mathematical differential equations.
Displayed as frames in a movie film, the book's front cover illustrated the diffusion of molecules from an initial state concentrated in the left half of a two-dimensional space to filling the whole space after seven frames.
Reif's work was a visualization of the second law of thermodynamics. Reif's movie frames led scientist
Stephen Wolfram to develop his "
New Kind of Science" using computational principles to derive the fundamental laws of physics.

On the back cover of the Reif book, the velocities of all the particles were reversed and the successive frames show the molecules making their way back into the left half. But they do not return to the exact original positions, due to "round-off errors" in the calculations.
In principle, if the computer could have unlimited information about the positions and velocities (an infinite number of significant figures), all the molecule positions in the past and the future could be determined by Newton's laws of motion (
F=ma, etc.). Classical physics is
deterministic.
In 1814,
Pierre-Simon Laplace wrote in the introduction to his
Essai philosophique sur les probabilités,
"We may regard the present state of the universe as the effect of its past and the cause of its future. An intellect which at a certain moment would know all forces that set nature in motion, and all positions of all items of which nature is composed, if this intellect were also vast enough to submit these data to analysis, it would embrace in a single formula the movements of the greatest bodies of the universe and those of the tiniest atom; for such an intellect nothing would be uncertain and the future just like the past would be present before its eyes."
LaPlace was extending an earlier idea of Newton's contemporary
Gottfried Leibniz, but it became famous as
Laplace's Demon, a key concept of strict physical
determinism.
In his latest book,
The Second Law, Stephen Wolfram says he spent fifty years trying to understand the deep physical significance of Reif's movie frames and the computations that produced them.
The great question for Wolfram, and for Reif, is this: if the equations of motion for microscopic collisions between gas particles are
reversible, why are the macroscopic properties of gases
irreversible, for example the entropy can only increase, never decrease, as the second law claims.
In a recent YouTube video, Wolfram described the problem,

And in his most recent book
The Second Law (p.219), Wolfram describes Fred's book that started his fifty-year quest to understand the second law as follows.
The Backstory of the Book Cover That Started It All
 What is the backstory of the book cover that launched my long journey with the Second Law? The book was published in 1965, and inside its front flap we find:
What is the backstory of the book cover that launched my long journey with the Second Law? The book was published in 1965, and inside its front flap we find:
The book covers
The movie strips on the covers illustrate the fundamental ideas of irreversibility and fluctuations by showing the motion of 40 particles inside a two-dimensional box. The movie strips were produced by an electronic computer programmed to calculate particle trajectories. (For details, see pp. 7, 24, and 25 inside the book.) The front cover illustrates the irreversible approach to equilibrium starting from the highly nonrandom initial situation where all the particles are located in the left half of the box. The back cover (read in the upward direction from bottom to top) illustrates the irreversible approach to equilibrium if, starting from the initial situation at the top of the front cover, all the particle velocities are reversed (or equivalently, if the direction of time is imagined to be reversed). The back-cover and front-cover movie strips together, read consecutively in the downward direction, illustrate a very large fluctuation occurring extremely rarely in equilibrium.
Wolfram designed the covers of his book to match the look of Fred's book, but with the computer calculations likely redone using his
Mathematica and
Wolfram Language tools, or perhaps the evolving hypergraphs of his cellular automata?

Is Physics Reversible or Irreversible?
The answer hinges on the question of fundamental
randomness.
Ludwig Boltzmann hypothesized there is some unknown process causing random behavior in the gas molecules that he called "molecular disorder" (
molekular ungeordnete).
Since Newton's microscopic laws of motion of the gas particles are completely deterministic and time reversible, the great question for the past one-hundred and fifty years is how macroscopically, the gas appears to be irreversible.
What can we say about the views of Fred Reif and Stephen Wolfram on the questions of randomness and reversibility? We can actually tell a lot by looking very carefully at the results of their computer calculations shown on the front and back covers of their books.
Here are the front cover and back cover movie strips side by side.
Reif Statistical Physics
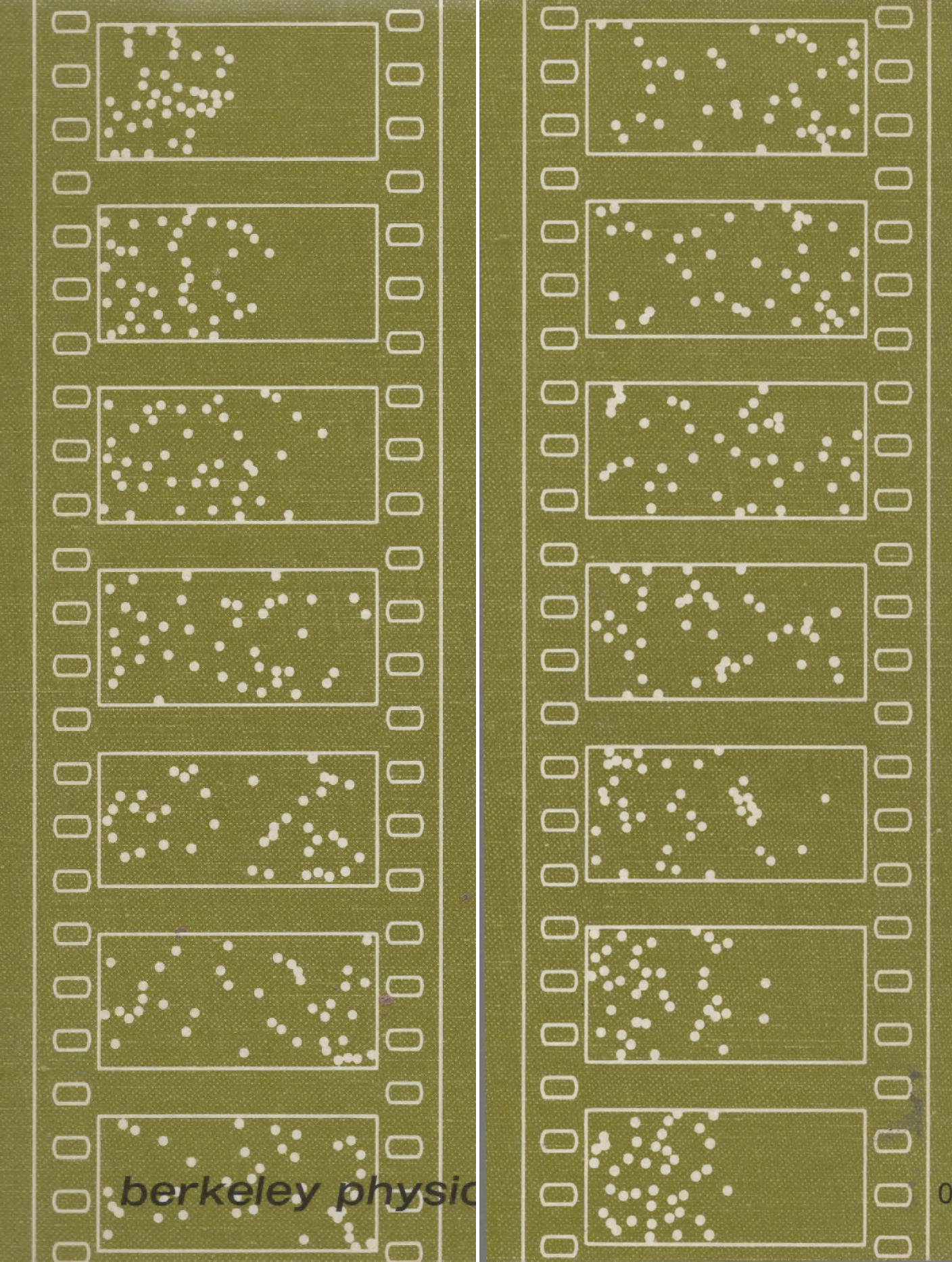
|
Wolfram The Second Law

|
Let's now carefully compare the starting frame on the front covers to the ending frame on the back covers. What can we say about the physics?
Both back covers start by reversing the velocities of the molecules in the last frame on the front cover. Wolfram shows that explicitly by reversing the little white arrows.
Let's look more closely at the starting and ending frames.
Both evolve back to the left half of the frames, as if time itself was being reversed.
But note that Reif's computations do not return each molecule back to its exact starting position, as do Wolfram's. Reif's calculations were done by Berni Alder on one of the most powerful computers in the 1960's, the Livermore Advanced Research Computer (LARC). Despite its power, the computer undoubtedly had roundoff errors, which caused the failure to return to their starting positions.
Wolfram's molecules start in a rigid grid pattern, initially four rows of three squares, perhaps an artifact of his cellular automata? Although the back cover unfortunately cuts off the bottom row of four molecules, the 12 upper molecules have returned to their exact original positions, as classical Newtonian physics predicts.
In Wolfram's terminology, classical physics is
computationally reducible.
Normal |
Teacher |
Scholar