A Book of Scandals
The
Information Philosopher has for years claimed to have
plausible solutions for many
great problems in philosophy and physics. We have called it a scandal that the world's educators have been teaching seriously outmoded ideas to young minds around the world.
The main scandal is academic philosophers and scientists teaching that the world is nothing but material and that physical events are completely
determined by "laws" of nature, that our bodies are machines and our
brains are computers that deny our
free will and our
creativity.
Information philosophy and science explains how
information structures are
created in the universe despite the second law of thermodynamics, which says that disorder or
entropy can only increase in a
closed thermodynamic system.

Click for the story of information creation
But the universe is not
closed, it is an
open thermodynamic system, in which the total
positive entropy or
disorder is increasing, but regions of "negative entropy" are also increasing, so information structures like our Sun and life on Earth are increasing the information content of the universe (the
Ergo), especially that part of information that is all of human knowledge (the
Sum).
Why do academic philosophers and scientists cling to failed ideas?
Philosophers prefer problems they have been taught to teach.
Scientists seek solutions that discover all-powerful new "laws of Nature."
Leading scientists have
interpreted quantum physics and
statistical thermodynamics, expressing their own doubts, so it is understandable that those less familiar with these complex subjects have fallen back on their traditional beliefs.
Philosophers
of science have been quick to exploit those doubts and to promote alternative theories that are not supported by evidence, but that appeal to our conventional beliefs.
Science journalists also find that speculative articles and books promoting these alternatives are
far more popular than serious attempts to teach these difficult subjects to the public.
The Two Things You Need to Know to End the Scandals
1. The standard theory of quantum mechanics is
correct. The many attempts to alter it or reinterpret it to restore
determinism have failed.
The
ontological chance discovered by
Albert Einstein in 1916, long before the
epistemological "uncertainty" principle of
Werner Heisenberg, is
real.
Chance drives the variation in living things that natural and intelligent selection choose between to give us
life, human beings, and our
knowledge of the universe.
Chance gives us the
alternative possibilities for acting that make us
free and
creative.
Einstein's Insight
In 1916
Albert Einstein found that when a photon is emitted by an atom, it must go off in a random direction. He called it "chance." Because he was a
determinist, he called it a "weakness in the theory." If the photon went in any preferred direction, the radiation field would depart from equilibrium, even permitting a perpetual motion machine, which he thought impossible.
2. The universe is
open and
expanding and
new information is
emerging at every moment.
Our futures are also
open.
If we have the will, we can
create new information structures that make
life more productive as well as more sustainable to protect our planet.
The origin of the universe was not "fine-tuned" to produce human life. Life (biology) cannot be
reduced to chemistry, which cannot be
reduced to a deterministic physics.
Boltzmann's Entropy, Eddington's Arrow, Layzer's Order
In 1872
Ludwig Boltzmann theorized that atoms or molecules of a gas would distribute themselves randomly throughout a closed container. If they all started in a corner, or were let out of a bottle, information about those initial positions would be lost. They would never return to those initial positions. The change is
irreversible. A few years later he modified his theory, saying particles would only
statistically never return. In infinite time, a finite number of particles could return, even more than once.
Boltzmann's theory allows us to calculate the
entropy increase when the volume available increases, and to calculate the loss in information.
In 1927
Arthur Stanley Eddington called the direction of entropy increase the "arrow of time." A few years later the expansion of the universe was discovered, and in 1935 Eddington suggested that if the expansion was faster than particles can redistribute themselves, particles could never return to their original distribution. Information was permanently lost.
In the 1970's
David Layzer said if the expansion is faster than particles can reach an equilibrium distribution, stable information structures can appear.
The mistaken idea in statistical physics that any particular distribution or arrangement of material particles has exactly the same information content as any other distribution is an anachronism from nineteenth-century statistical physics.
 |
 |
 |
| Hemoglobin |
Diffusing |
Completely Mixed Gas |
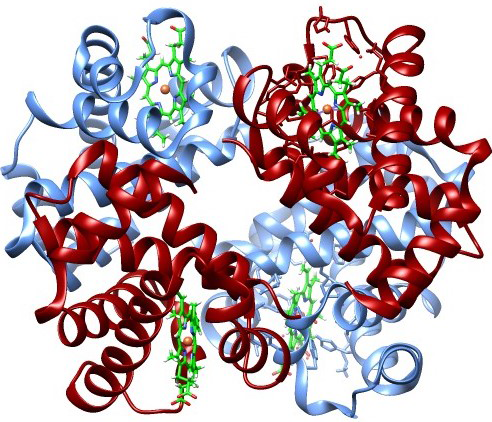
If we measure the positions in
phase space of all the atoms in a hemoglobin protein, we get a certain number of bits of data (the x, y, z, v
x, v
y, v
z values). If the chemical bonds are all broken allowing atoms to diffuse, or the atoms are completely randomized into an equilibrium gas with maximum entropy, we get different values, but the same amount of data. Does this mean that any particular distribution has exactly the same information?
This led many statistical physicists to claim that information is the same wherever the particles are.
Macroscopic information is not lost, it just becomes
microscopic information that can be completely recovered if the motions of every particle could be reversed.
Reversibility allows all the gas particles to go back inside the bottle.
But the
information in the hemoglobin is much higher and the disorder (entropy) near zero. A human being is not just a "bag of chemicals," despite plant biologist
Anthony Cashmore. Each atom in hemoglobin is not merely in some volume limited by the uncertainty principle ℏ
3, it is in a specific quantum cooperative relationship with its neighbors that support its biological function. These precise positional relationships make a passive linear protein into a properly folded active enzyme. Breaking all these quantum chemical bonds destroys
life.
Boltzmann's formula for the entropy is the logarithm of the number W of possible distributions that produce the same macroscopic properties.
S = k ln W
When gas particles can go anywhere in a container, the number of possible distributions is enormous and entropy is maximal. When atoms are bound to others in the hemoglobin structure, the number of possible distributions is essentially
1, and the logarithm of
1 is
0!
Even more important, the parts of every living thing are
communicating information - signaling - to other parts, near and far, as well as to other living things. Information communication allows each living thing to maintain itself in a state of
homeostasis, balancing all matter and energy entering and leaving, maintaining all vital functions. Statistical physics and chemical thermodynamics know nothing of this biological information.
One Book and Many
In our initial "book of scandals" we present brief summaries of the major scandals/problems and their solutions in terms of the
creation of information structures, their
processes,
functions, and, most important, their
communications with one another.
Each scandal will become its own book, if the author lives long enough in the current age of the coronavirus (he will be 90 in June 2026). Many are already sections, or at least pages, in this I-Phi website, which has been a work in progress for nearly three decades.
Deep insight into the informational nature of reality will can show thinkers young and old why they are
creators of our open future as human beings.
Normal |
Teacher |
Scholar